Nordlys® - It's The One For Acne
In recent years, energy-based systems treating acne have made a big splash in the aesthetics industry. It makes sense, since acne is the most common skin condition in the United States, affecting up to 50 million Americans annually.1
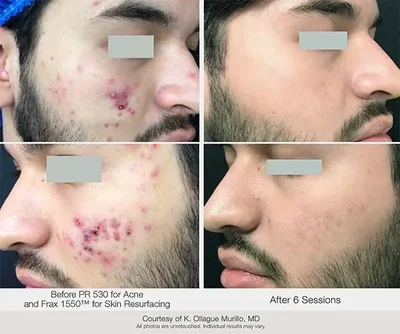 But, buyer beware! Before investing in a new energy-based device to address acne, you should research whether it’s clinically proven to address your business and patients’ needs. One versatile device that is FDA cleared and Health Canada licensed to treat inflammatory acne vulgaris, address redness, and smooth and resurface skin, is the Nordlys® multi-application platform.2-3
But, buyer beware! Before investing in a new energy-based device to address acne, you should research whether it’s clinically proven to address your business and patients’ needs. One versatile device that is FDA cleared and Health Canada licensed to treat inflammatory acne vulgaris, address redness, and smooth and resurface skin, is the Nordlys® multi-application platform.2-3
The Nordlys system uses Selective Waveband Technology (SWT®) IPL (PR 530) to address inflammatory acne vulgaris and redness, and its 1550 and 1940 non-ablative fractional lasers resurface skin at both deep and shallow levels, respectively.
Some recent energy-based device market entrants, such as UltraClear (2021) and AviClear (2022) address either active acne or acne scars/skin resurfacing, but not both. In contrast, the versatile Nordlys system technologies include FDA-cleared treatments to address active acne, eliminate redness, and resurface skin textural irregularities, three common skin issues in acne patients.4 In fact, the Nordlys system is FDA-cleared to treat 22 indications*, including vascular conditions, pigmentation, and hair removal.
If you are seeking a device that extends both treatments and value for your practice, the choice is clear! It’s the one - the Nordlys system.
 Japan
Japan Korea
Korea China
China